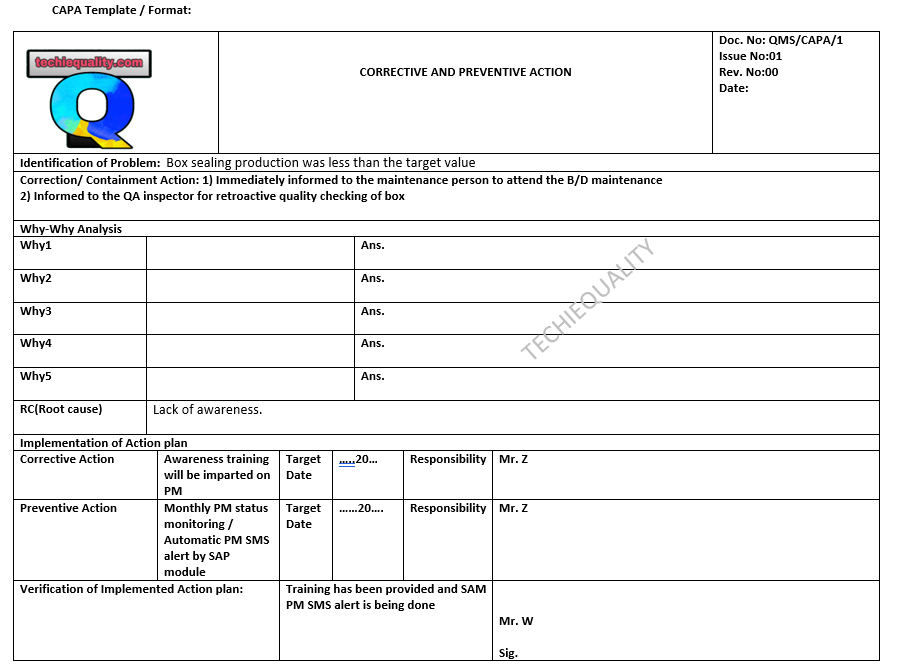
why why analysis methodology | 5-why analysis step by step guide:
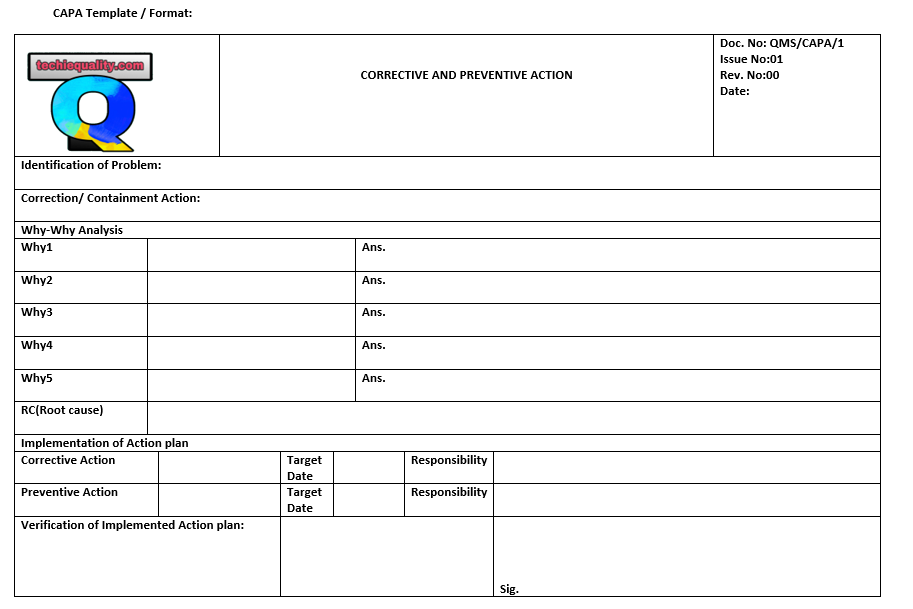
Hello readers! Today we will discuss “why why analysis methodology”. How effectively you can do the 5-why analysis. This analysis is the common tool used in the industry to find out the root cause of the problem but most of the people make mistakes during why-why analysis. So we will talk about the step-by-step methodology of why-why analysis. The why-why analysis is one of the inputs for executing the 8D report, CAPA (Corrective action and preventive action), and RCA (Root Cause analysis). Click on the below-given link to download the 8D and CAPA format.
Download 8D Format , CAPA Format & 5W1H Format.
You may like to read similar topics;
Root Cause Analysis | 8 Steps of RCA
8D Report | Free Download of 8D Template | Format
Free Template / Format (QA/ QC)
Decision of process capability analysis |Download Format
How to measure process capability (Cp & Cpk)? Download the Excel Template.
How to measure process performance (Pp & Ppk)?
why why analysis methodology:
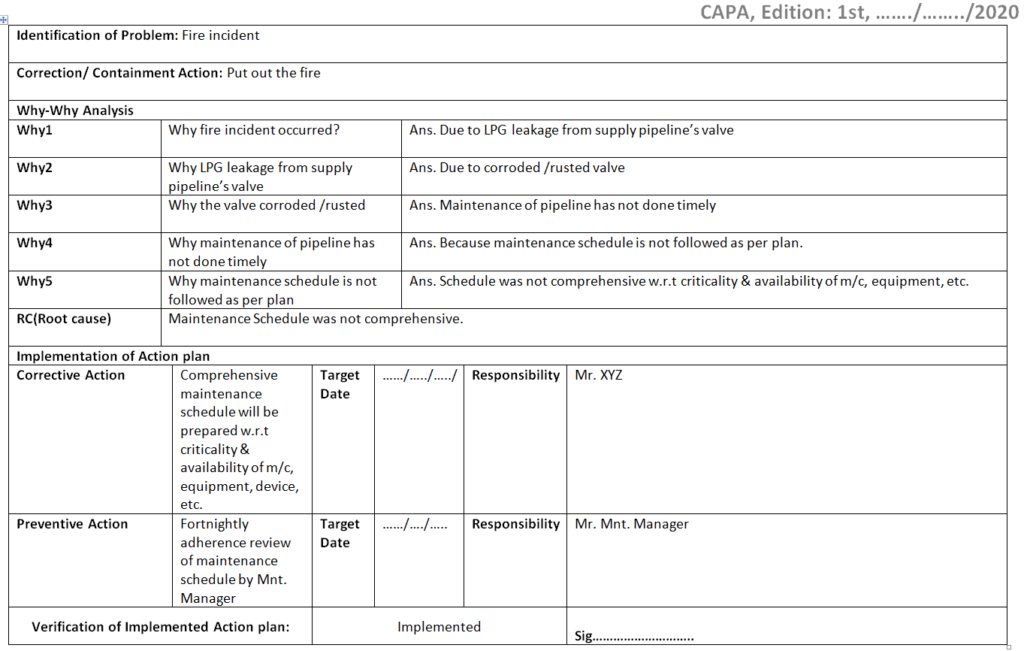
Step-1: Identify the Problem.
Step-2: Understand the Problem.
Step-3: CFT team formation.
Step-4: Identify the Potential cause
Step-5: Why-why analysis.
Understanding the why-why analysis methodology by example:
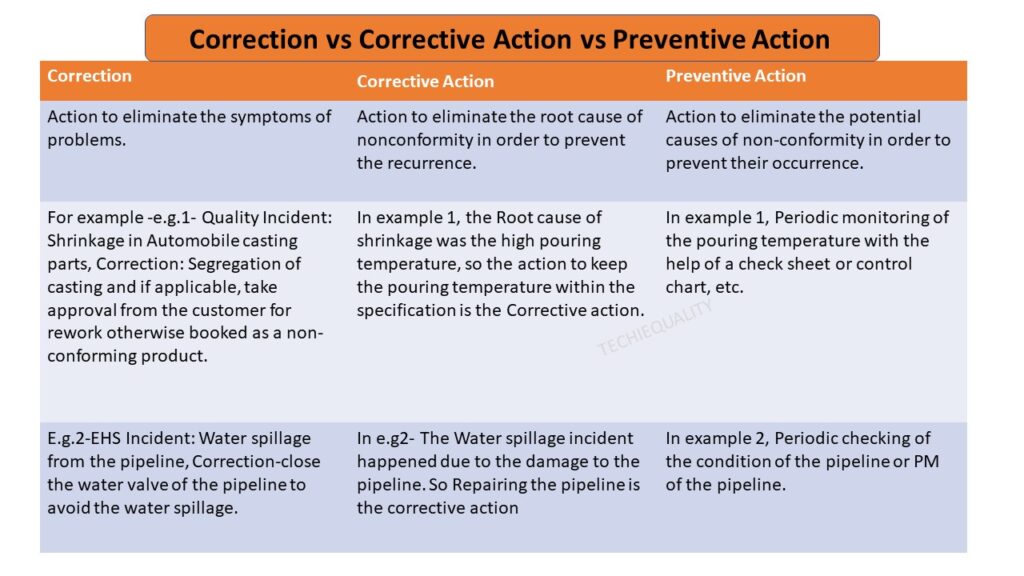
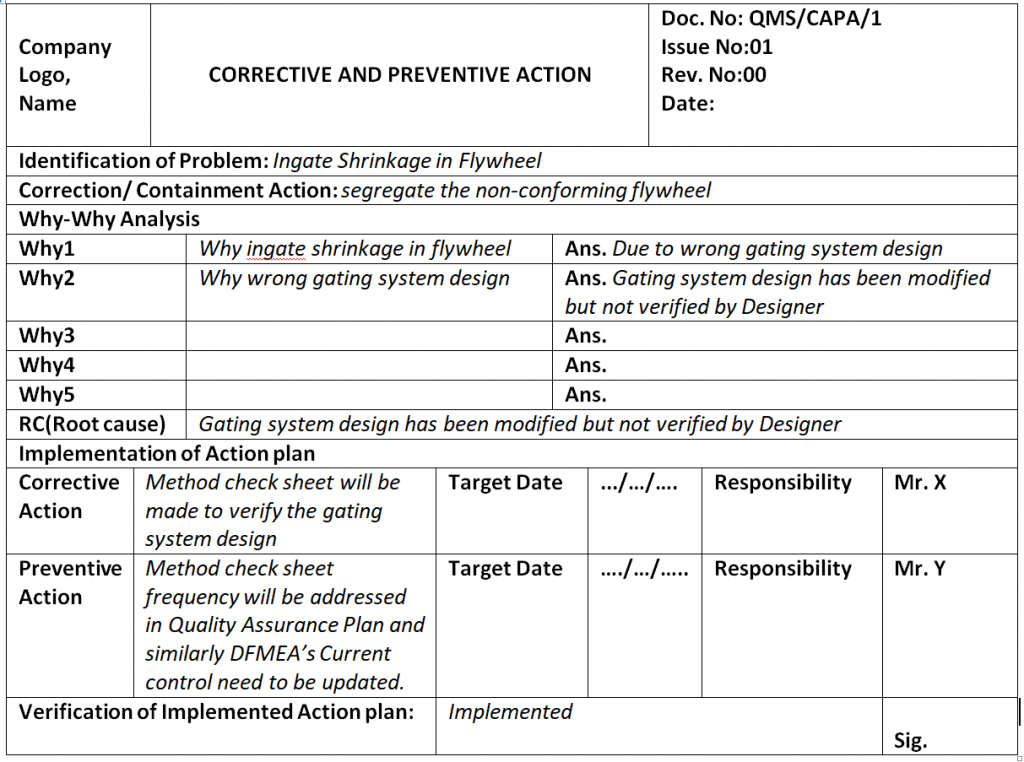
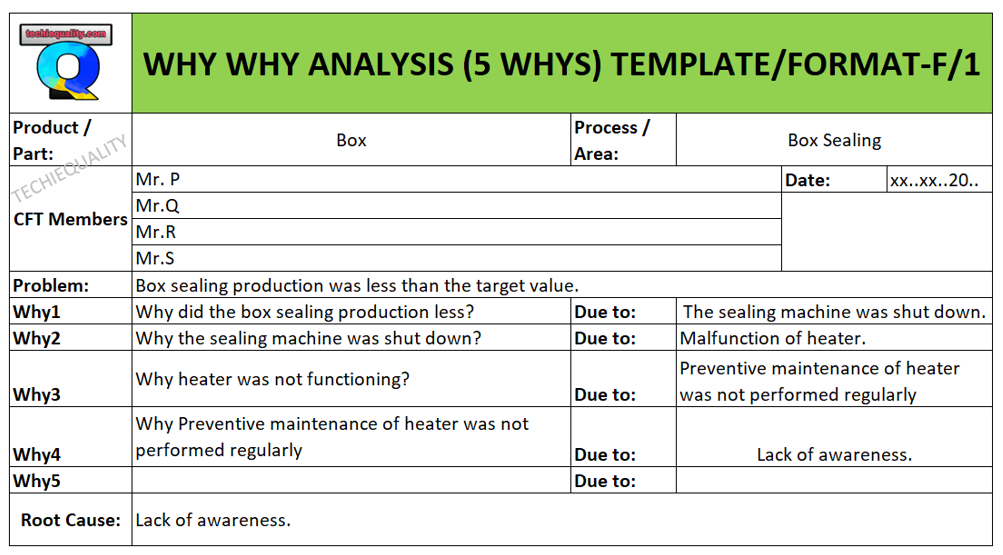
Let Part number 123 find ingate shrinkage after the shot-blasting operation. So this case process-QA engineer started the why-why analysis by using the above methodology.
Identify the Problem:
At the shot-blasting operation, a process-QA engineer identified that two number casting got rejected due to ingate shrinkage.
Understand the Problem:
Understanding the problem means technically knowing what exactly is it? How is it occurring? (potential cause). In the above example, we are supposed to have knowledge on “what is shrinkage?
CFT team formation:
If you think that the problem may be due to issues with the melting parameter, moulding parameter, or design. Then you have to create a CFT team considering with the above departments.
Identify the Potential cause:
Now initially individual members will identify the potential cause. And then combine all members of the CFT team will identify the potential cause. Let’s see, in case of a shrinkage problem what would be the final list of potential cause.
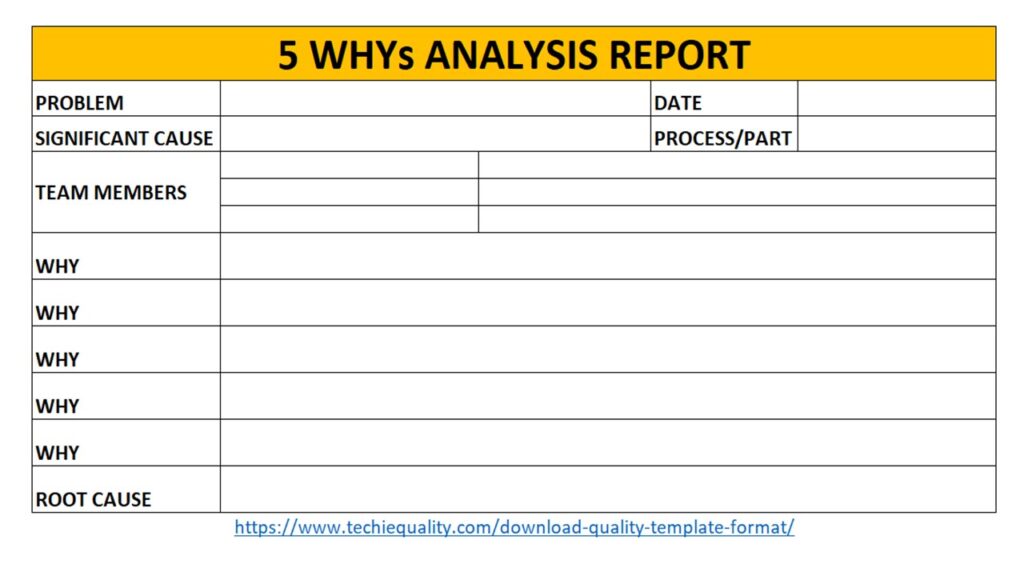
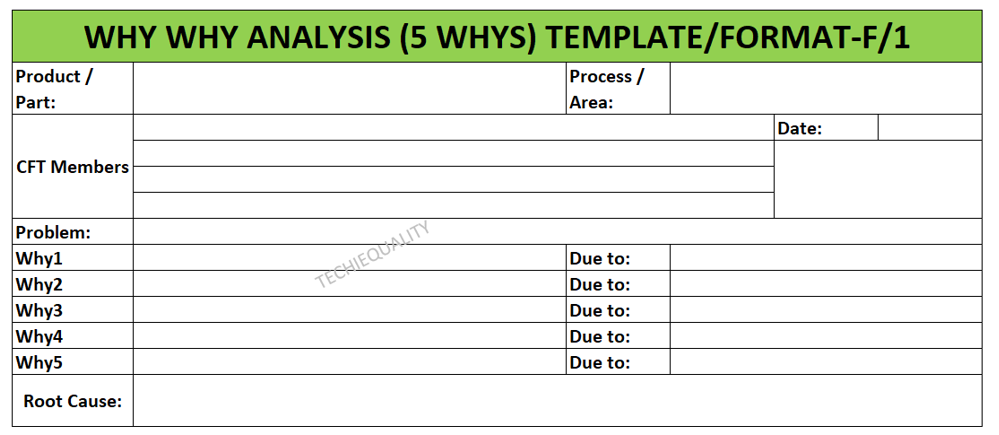
Why-why analysis:
In this section, we are supposed to identify the significant cause of the potential cause. We have already identified the potential cause with the help of the CFT team and represented it through cause and effect diagram.
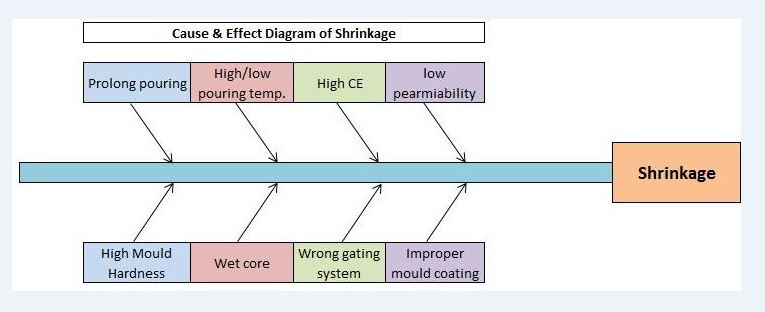
To identify the significant cause from the above C&E Diagram, we need to use the methodology like brainstorming method, validation method, and hypothesis testing method. With the help of the brainstorming and validation method, we found that the “wrong gating system” is the significant cause. So now we will do the Why-why analysis by CFT team members.
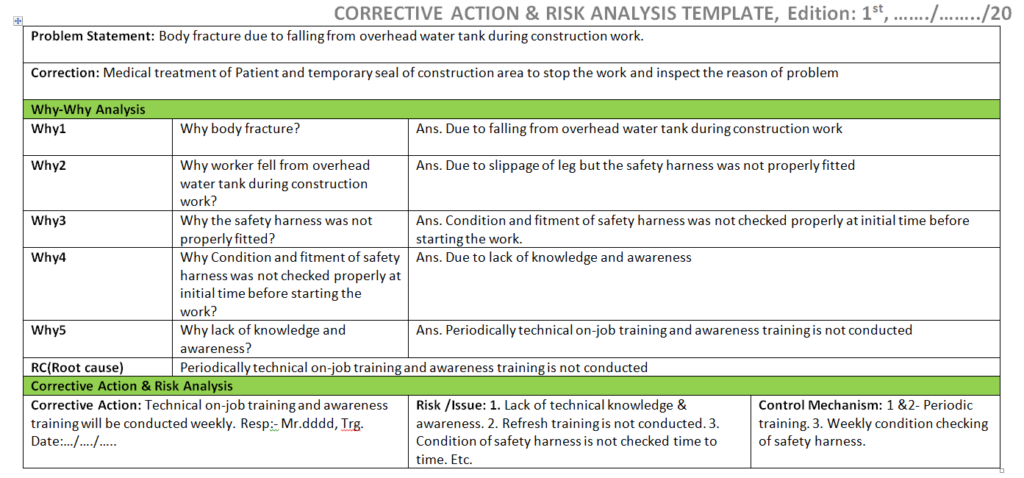
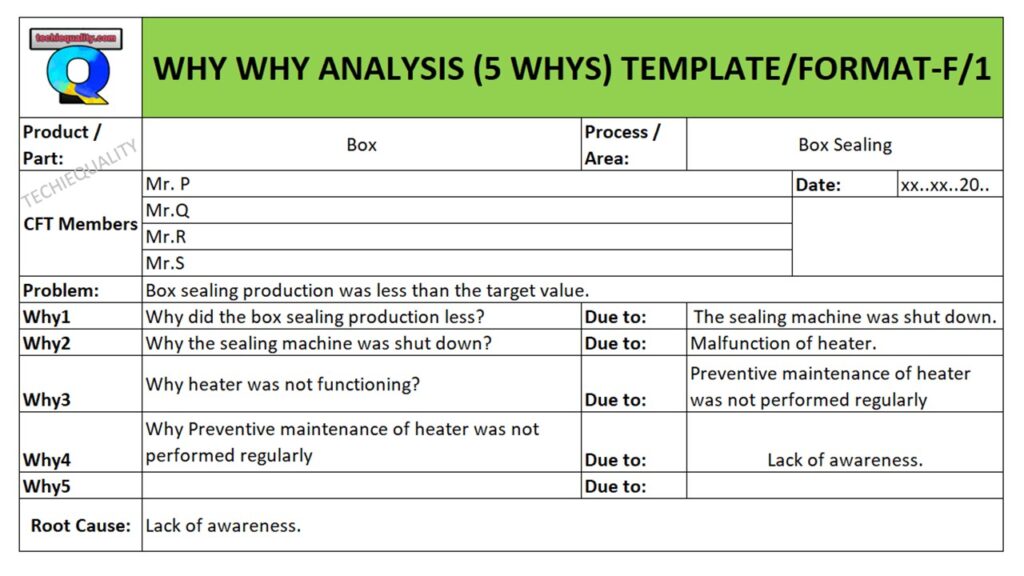
Problem: Wrong gating system.

Why wrong gating system?
Ans.: gating system was modified

Why gating system was modified?
Ans.: Gating system was modified w.r.t customer complaint but gating system was not validated before mass production.

Why gating system was not validated?
Ans.: There was no time to validate the part because production planning was scheduled without prior consideration of tool validation

RC: Improper Production planning and Schedule.
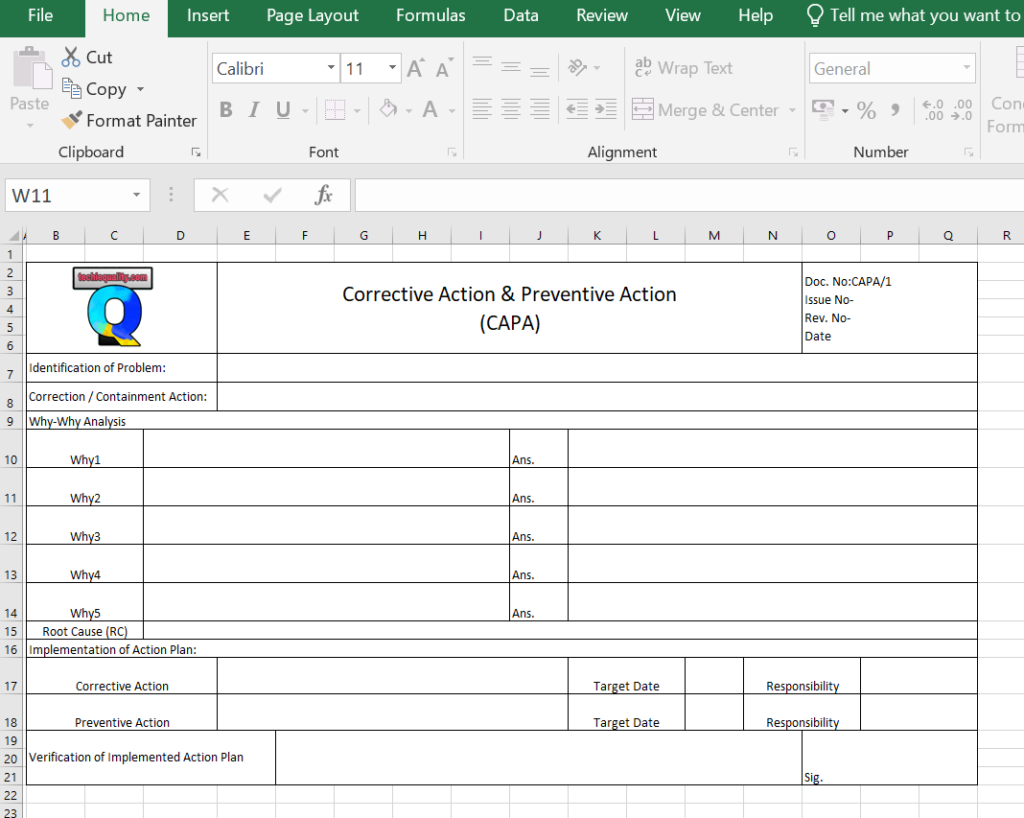
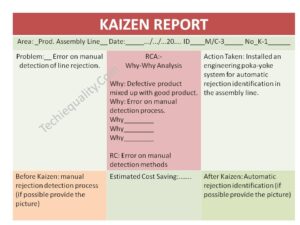
Once you completed the analysis you can document these in a 5-whys analysis template.
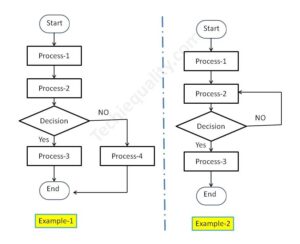
Funnel Approach of Why-Why Analysis:
Once you identify the potential cause then we could also use the funnel approach to identify the Root cause by 5-why analysis.
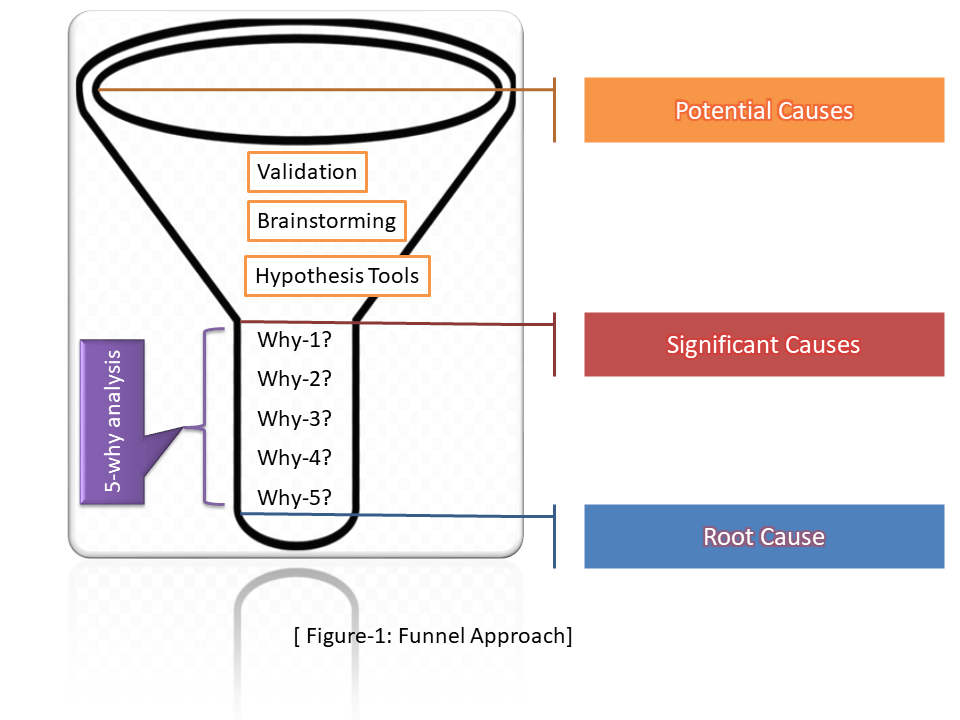
In the above funnel diagram, we have mentioned three basic stages and applied some tools/ methods/ testing like validation, brainstorming, hypothesis testing, etc. to identify the significant cause in stage-2. and then applied the 5-why analysis to detect the Root cause in stage-3. In this way, we can use this approach to identify the RC.
Why-Why Analysis Methodology to Identify the Multiple Root Cause:
In the above section, we learned the two different methodologies for why-why analysis. and in both case, we found a single root cause. we know that theoretically there will be a single root cause of a problem but technically there will be a minimum one root cause of a problem. So here we have mentioned in the below figure for the methodology to identify the multiple root causes.
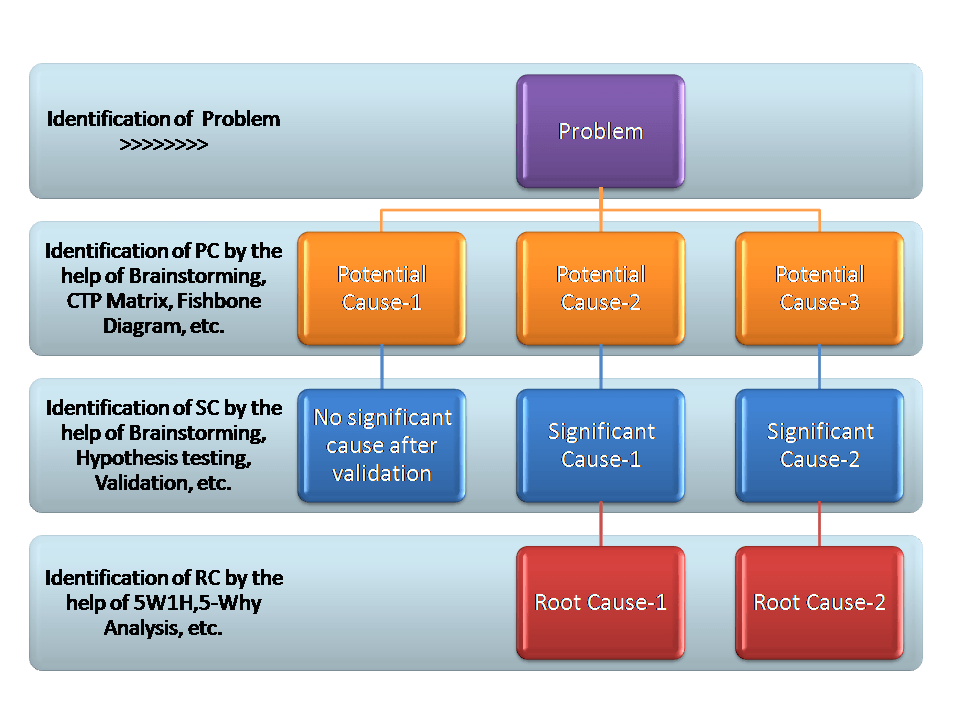
Step-1: Identification and Understanding of the Problem
Step-2: CFT Team Formation and list up the Potential Causes by using tools like Brainstorming, CTP Matrix, Fishbone Diagram, etc.
Step-3: Significant Cause identification with the help of Brainstorming, Hypothesis Testing, Validation, etc.
Step-4: Identification of Root Cause by why-why/ 5W1H analysis of each individual Significant Cause.
Similar Post:
8D Report Example | Download Case Study Report:
8D Report | Free Download of 8D Template | Format
7QC Tools for Problem Solving | What are 7 QC Tools
How to Plot Pareto Chart in Excel ( with example)
How to plot Histogram in Excel (Step by step guide with example)
More on TECHIEQUALITY
Thank you for reading…Keep visiting Techiequality.Com
Popular Post:
Shanti Gopal Pradhan is an experienced professional in Quality Management Systems, QA, Operations, Business Excellence, and Process Improvement. He has strong expertise in international standards including IATF 16949, ISO 9001, ISO 14001, ISO 45001, and ISO 17025, along with methodologies such as TQM, TPM, and Six Sigma.
He holds a degree in Mechanical Engineering along with an MBA, combining strong technical acumen with strategic business insight, he is a Certified Internal Auditor, Lead Auditor, and Six Sigma Black Belt, with a proven track record in driving quality transformation and operational excellence.